Neuroprotective Therapies
Neuroprotective Therapies: Mechanisms, Agents, and Clinical Applications
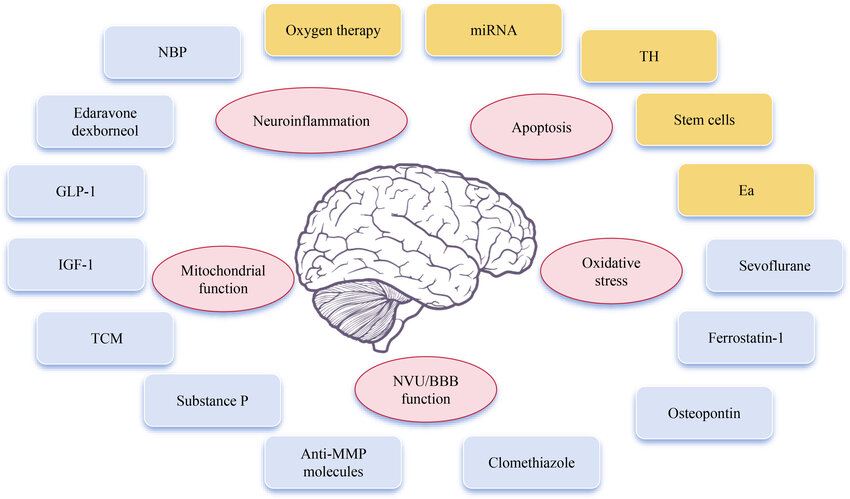
Neuroprotective therapies aim to preserve neuronal structure and function, mitigate secondary damage, and slow progression in neurological disorders such as neurodegenerative diseases (e.g., Alzheimer’s, Parkinson’s), stroke, traumatic brain injury (TBI), and neurotoxin exposure. These therapies target shared pathological mechanisms, including oxidative stress, excitotoxicity, inflammation, and apoptosis.
Key Mechanisms of Neuroprotection
- Oxidative Stress Mitigation:
- Neutralizes reactive oxygen species (ROS) via antioxidants (e.g., vitamin E, coenzyme Q10) and activation of the Nrf2 pathway, which upregulates detoxifying genes.
- Excitotoxicity Inhibition:
- Blocks glutamate overactivation using NMDA receptor antagonists (e.g., memantine) to prevent calcium overload and neuronal death.
- Anti-Apoptotic Strategies:
- Endoplasmic Reticulum (ER) Stress Modulation:
- Activates unfolded protein response (UPR) pathways (PERK, ATF6, XBP1) to restore proteostasis.
- Autophagy Regulation:
- Fine-tuned autophagy clears toxic protein aggregates (e.g., amyloid-β, tau) while avoiding excessive degradation.
- Anti-Inflammatory Actions:
- Reduces neuroinflammation via microglial modulation (e.g., NSAIDs, resolvins like RvD1).
Therapeutic Agents and Approaches
| Agent/Approach | Mechanism | Clinical Applications |
|---|---|---|
| Lithium | Inhibits GSK-3β, upregulates BDNF, reduces tau hyperphosphorylation. | Alzheimer’s, bipolar disorder. |
| Erythropoietin | Reduces hypoxia-induced glutamate toxicity. | Stroke, TBI. |
| Therapeutic Hypothermia | Lowers metabolic demand, reduces ROS and inflammation. | Neonatal encephalopathy, cardiac arrest. |
| Resolvins/Neuroprotectins | Derived from omega-3 fatty acids (DHA/EPA); resolve inflammation. | Alzheimer’s, retinal degeneration. |
| Nrf2 Activators | Boost antioxidant defenses (e.g., sulforaphane). | Parkinson’s, ALS. |
| Trophic Factors | Promote neuronal survival (e.g., BDNF, GDNF). | ALS, spinal cord injury. |
| Natural Compounds | Curcumin, resveratrol modulate multiple pathways. | Neurodegenerative diseases, stroke. |
Clinical Applications
- Acute Conditions:
- Stroke: Hypothermia, erythropoietin, and NMDA antagonists limit infarct size.
- TBI: Antioxidants (e.g., NAC) and anti-inflammatory agents reduce secondary damage.
- Chronic Neurodegeneration:
- Alzheimer’s: Lithium, resolvins, and autophagy enhancers target amyloid-β and tau.
- Parkinson’s: Nrf2 activators and GDNF protect dopaminergic neurons.
- ALS: Trophic factors (IGF-1) and caspase inhibitors delay motor neuron loss.
Challenges
- Timing: Neuroprotection is most effective when applied early in disease progression.
- Complex Pathways: Overlapping mechanisms (e.g., ER stress and autophagy) require multitarget therapies.
- Clinical Translation: Many agents show promise in preclinical models but fail in trials due to bioavailability or off-target effects.
Future Directions
- Combination Therapies: Pairing antioxidants with anti-excitotoxics (e.g., memantine + vitamin E).
- Gene-Based Approaches: CRISPR/Cas9 to enhance endogenous neuroprotective pathways (e.g., Nrf2).
- Biomarkers: Identifying early indicators (e.g., tau levels) to guide intervention timing.
- Natural Products: Optimizing bioavailability of compounds like curcumin for clinical use.
Conclusion
Neuroprotective therapies focus on halting disease progression by targeting oxidative stress, inflammation, and apoptosis. While current agents like lithium and resolvins show promise, challenges in timing and complexity necessitate innovative strategies. Future advances in personalized medicine and combination therapies may unlock transformative outcomes.