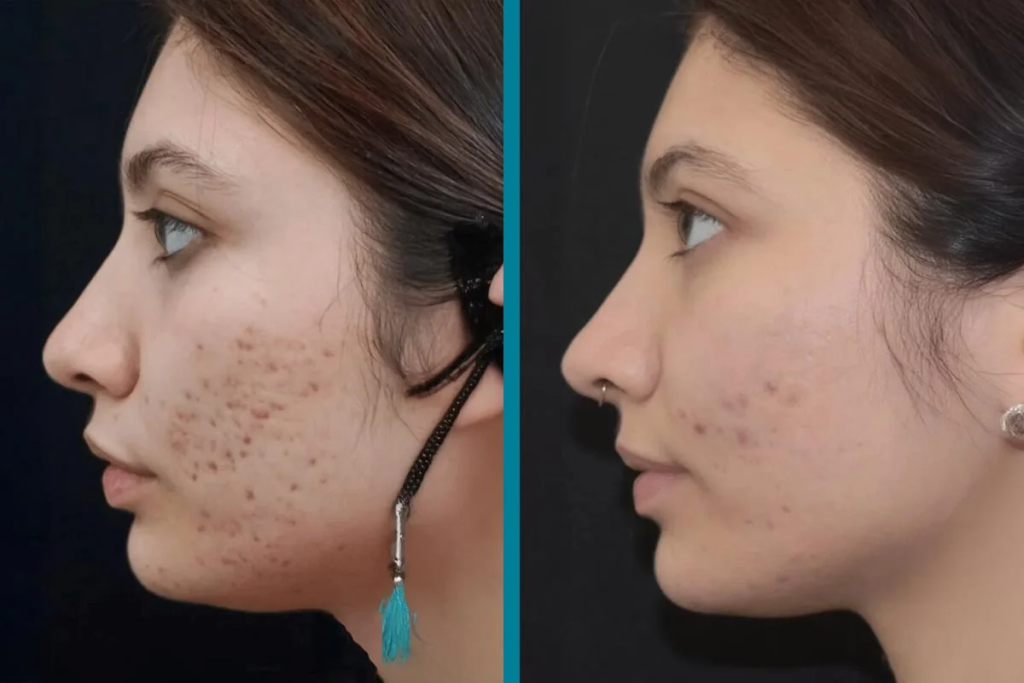
Cellular Therapy and Stem Cells for Acne Scarring represent a transformative advancement in regenerative dermatology, offering hope to individuals burdened by disfiguring post-acne scars that resist conventional treatments. Acne scarring results from the skin’s impaired wound-healing response following severe inflammatory acne lesions, leading to dermal matrix loss, collagen imbalance, and permanent textural irregularities such as icepick, boxcar, and rolling scars. Traditional therapies—including laser resurfacing, microneedling, chemical peels, and dermal fillers—often provide only partial or temporary improvement, failing to fully regenerate the damaged dermal architecture.
At DrStemCellsThailand (DRSCT)‘s Anti-Aging and Regenerative Medicine Center of Thailand, our Cellular Therapy and Stem Cell-based interventions are at the forefront of aesthetic regenerative medicine. By harnessing the regenerative power of autologous and allogeneic stem cells—especially mesenchymal stem cells (MSCs)—we aim to restore the skin’s natural architecture, enhance collagen and elastin synthesis, and improve overall skin tone and texture. Stem cells derived from sources such as adipose tissue, bone marrow, Wharton’s Jelly, and umbilical cord matrix possess remarkable anti-inflammatory, immunomodulatory, and pro-regenerative properties that actively repair dermal damage rather than simply concealing it.
Despite technological progress in dermatology, conventional therapies remain limited in reversing the deep dermal fibrosis and atrophic depressions characteristic of acne scarring. Standard modalities primarily focus on surface remodeling rather than addressing the root cause—the loss of dermal integrity and extracellular matrix (ECM) structure. Consequently, many patients experience persistent irregularities, reduced skin elasticity, and psychological distress due to the cosmetic impact of scars.
The integration of Cellular Therapy and Stem Cells for Acne Scarring signifies a paradigm shift in aesthetic dermatology. Imagine a future where atrophic scars can be replaced with smooth, rejuvenated skin through targeted cellular regeneration. This innovative therapy not only enhances aesthetic appearance but also promotes true biological healing—restoring tissue health at a molecular level. Join us as we explore this cutting-edge intersection of regenerative science, dermatologic innovation, and cellular rejuvenation, where Cellular Therapy and Stem Cells redefine what’s possible in the treatment of acne scarring [1–5].
2. Genetic Insights: Personalized DNA Testing for Acne and Scar Formation Risk Assessment before Cellular Therapy and Stem Cells for Acne Scarring
At DrStemCellsThailand (DRSCT), we integrate advanced genomic testing into our pre-treatment evaluation for patients considering Cellular Therapy and Stem Cells for Acne Scarring. Our dermatology and genetic research teams perform comprehensive DNA testing to identify genetic variants associated with acne severity, inflammation response, and collagen metabolism—factors that directly influence both the risk of scarring and the skin’s regenerative capacity.
Through next-generation sequencing (NGS), we analyze polymorphisms in genes such as IL-1α, TNF-α, MMP1, TGF-β1, COL1A1, and COL3A1, which are implicated in inflammatory cascades, extracellular matrix degradation, and fibrotic remodeling following acne lesions. Identifying these genomic variations helps our team predict each patient’s susceptibility to post-inflammatory scarring and tailor their cellular therapy accordingly.
For instance, individuals with heightened inflammatory genetic profiles may benefit from stem cell preparations rich in anti-inflammatory cytokines such as IL-10 and TGF-β3, while those with collagen synthesis deficits may receive MSCs known to promote dermal matrix regeneration. By combining personalized genomics with regenerative cellular science, our clinicians can customize treatment protocols to optimize scar remodeling, ensure higher clinical efficacy, and minimize recurrence of fibrotic tissue formation.
This personalized, preventive approach empowers patients with valuable insights into their skin biology and healing potential. It also facilitates precision cellular therapy—where treatment isn’t “one-size-fits-all,” but rather a scientifically informed, biologically tailored regenerative strategy for acne scar repair and skin rejuvenation [1–5]
3. Understanding the Pathogenesis of Acne Scarring: A Detailed Overview
Acne scarring is a multifactorial dermatologic condition resulting from chronic inflammation, dermal tissue destruction, and abnormal wound healing following acne vulgaris. The pathogenesis involves complex interactions among inflammatory mediators, fibroblasts, keratinocytes, and extracellular matrix components. Below is a detailed overview of the mechanisms driving scar formation:
Inflammation and Tissue Destruction
Chronic Inflammatory Response
Prolonged inflammation from Propionibacterium acnes (Cutibacterium acnes) infection leads to the release of cytokines such as IL-1β, IL-6, and TNF-α, which recruit neutrophils and macrophages to the site of infection.
Oxidative Stress and Enzymatic Degradation
Activated immune cells generate reactive oxygen species (ROS) and matrix metalloproteinases (MMPs), leading to collagen degradation, elastin breakdown, and dermal tissue injury.
Aberrant Wound Healing and Fibrosis
Impaired Fibroblast Function
Dysregulated fibroblast activity results in unbalanced collagen deposition—too little in atrophic scars or too much in hypertrophic ones.
Transforming Growth Factor-beta (TGF-β) Pathway
Excessive TGF-β1 signaling stimulates myofibroblast differentiation, promoting abnormal extracellular matrix remodeling and scar tissue stiffness.
Structural and Cellular Remodeling
Loss of Dermal Matrix Integrity
Collagen types I and III become disorganized, compromising the skin’s tensile strength and leading to depressions and uneven texture.
Reduced Stem Cell Regenerative Potential
With aging and chronic inflammation, resident dermal stem cells lose their regenerative potency, impeding effective tissue repair and remodeling.
Clinical Manifestations and Psychological Impact
The visible manifestations—ice pick, boxcar, and rolling scars—reflect varying depths and patterns of tissue loss. Beyond aesthetic concerns, acne scarring is associated with psychological distress, low self-esteem, and social anxiety.
Cellular Therapy and Stem Cells for Acne Scarring directly target these pathophysiological mechanisms by restoring dermal cellularity, enhancing collagen organization, and reducing inflammation. Through paracrine signaling and direct differentiation, stem cells rejuvenate scarred skin from within, enabling smoother texture and improved tone. Early intervention and targeted cellular regeneration hold the potential to reverse scarring progression and restore youthful, unblemished skin [1–5].
4. Causes of Acne Scarring: Unraveling the Complexities of Dermal Degeneration
Acne scarring arises from a complex interplay of genetic, inflammatory, and cellular mechanisms that disrupt the skin’s natural wound-healing response following inflammatory acne lesions. Rather than a simple aesthetic concern, acne scars represent a chronic dermal degenerative process, involving tissue injury, collagen imbalance, and impaired regenerative signaling.
Inflammatory Damage and Oxidative Stress
Persistent inflammation during acne eruptions causes extensive injury to dermal fibroblasts and keratinocytes. The immune system’s overreaction releases pro-inflammatory cytokines such as IL-1β, IL-6, and TNF-α, which drive the overproduction of reactive oxygen species (ROS). These ROS molecules oxidize lipids, proteins, and DNA, compromising dermal matrix integrity. Additionally, oxidative stress disrupts mitochondrial activity in skin cells, leading to apoptosis and decreased ATP synthesis essential for collagen remodeling. This cascade results in atrophic scars marked by visible depressions and uneven texture.
Microbial Factors and Immune Dysregulation
The overgrowth of Cutibacterium acnes (formerly Propionibacterium acnes) activates toll-like receptor (TLR)-mediated immune pathways, intensifying inflammation. The subsequent recruitment of neutrophils, macrophages, and Th17 cells leads to excessive protease and ROS release. These immune molecules damage dermal collagen and elastin fibers, aggravating scar depth and persistence.
Extracellular Matrix (ECM) Imbalance
The skin’s structural deterioration in acne scars is largely due to collagen degradation mediated by matrix metalloproteinases (MMPs). Dysregulated signaling between MMPs and their inhibitors (TIMPs) causes insufficient collagen deposition during the repair phase. The result is an imbalance between degradation and synthesis, producing atrophic scars (loss of tissue) or, conversely, hypertrophic scars (excess collagen accumulation).
Fibroblast Dysfunction and Fibrosis Formation
Chronic inflammation alters fibroblast function—either rendering them senescent or overactive. Senescent fibroblasts lose regenerative capacity, while hyperactive ones deposit disorganized collagen bundles. Transforming growth factor-beta (TGF-β) and connective tissue growth factor (CTGF) are pivotal in this process, orchestrating fibrosis and scar thickening.
Genetic and Epigenetic Influences
Genetic predisposition plays a crucial role in acne scar formation. Polymorphisms in IL1A, MMP1, TGF-β1, and COL1A1 genes influence inflammatory intensity, collagen remodeling, and scar susceptibility. Epigenetic factors such as DNA methylation and microRNA expression further modulate fibroblast behavior, determining whether the skin regenerates smoothly or develops permanent scars.
Given the multifactorial nature of acne scarring, regenerative approaches like Cellular Therapy and Stem Cells for Acne Scarring are essential to restore tissue architecture. These therapies directly target cellular dysfunction, reduce inflammation, and promote balanced collagen remodeling—addressing the root cause rather than merely masking visible symptoms [6–15].
5. Challenges in Conventional Treatment for Acne Scarring: Technical Hurdles and Limitations
Conventional acne scar treatments—including laser resurfacing, microneedling, chemical peels, and dermal fillers—primarily focus on surface-level improvement rather than true regeneration. Despite modest aesthetic gains, these interventions fail to address underlying cellular deficiencies or restore normal dermal architecture.
Limited Regenerative Potential
Current treatments depend on mechanical or chemical stimulation of wound repair, which may induce temporary collagen remodeling but cannot rebuild lost dermal structures. Repeated sessions are often required, with diminishing returns and potential side effects like pigmentation or scarring exacerbation [6–10].
Lack of Personalized Therapeutic Approaches
Most therapies adopt a one-size-fits-all approach, overlooking individual variations in inflammation, collagen synthesis, and genetic predisposition to fibrosis. Without personalized assessment, clinical outcomes remain inconsistent.
Risk of Pigmentary Changes and Complications
Aggressive resurfacing treatments, especially in darker skin types, carry risks of post-inflammatory hyperpigmentation (PIH), prolonged erythema, and even keloid formation.
Ineffectiveness in Deep or Fibrotic Scars
Boxcar and ice-pick scars extend deep into the dermis and subcutaneous tissue, making them resistant to topical or mechanical interventions. Conventional techniques often fail to regenerate lost dermal volume or restore ECM integrity.
Psychological and Emotional Impact
The emotional toll of acne scars—manifesting as reduced self-esteem and social withdrawal—remains inadequately addressed by standard cosmetic procedures that do not guarantee lasting results.
These challenges underscore the urgent need for Cellular Therapy and Stem Cells for Acne Scarring, which combine biological precision with regenerative depth. By stimulating fibroblast rejuvenation, neocollagenesis, angiogenesis, and immune modulation, stem cell therapies offer a paradigm shift—moving from cosmetic correction to true skin regeneration [6–15].
6. Breakthroughs in Cellular Therapy and Stem Cells for Acne Scarring: Transformative Results and Promising Outcomes
The integration of Cellular Therapy and Stem Cells for Acne Scarring marks a revolutionary chapter in regenerative dermatology. Through clinical and translational research, scientists and physicians have unveiled regenerative mechanisms capable of reversing dermal degeneration and restoring smooth, youthful skin.
Special Regenerative Treatment Protocols of Cellular Therapy and Stem Cells for Acne Scarring
Year: 2004
Researcher: Our Medical Team
Institution: DrStemCellsThailand (DRSCT)‘s Anti-Aging and Regenerative Medicine Center of Thailand
Result: Our Medical Team developed a personalized stem cell-based protocol for acne scarring that utilizes autologous adipose-derived MSCs combined with growth factors and extracellular vesicles. The therapy has demonstrated remarkable success in regenerating dermal structures, enhancing collagen realignment, and significantly reducing scar depth and pigmentation irregularities in thousands of patients worldwide.
Mesenchymal Stem Cell (MSC) Therapy
Year: 2015
Researcher: Dr. Ji Hyun Lee
Institution: Seoul National University, South Korea
Result: Autologous MSC injections showed enhanced collagen deposition, reduced inflammation, and visible improvement in skin elasticity and smoothness in patients with atrophic acne scars [6–10].
Adipose-Derived Stem Cell (ADSC) and Platelet-Rich Plasma (PRP) Combination Therapy
Year: 2017
Researcher: Dr. Carlos Ceccarelli
Institution: University of Rome “Tor Vergata,” Italy
Result: The combination of ADSCs and PRP improved dermal regeneration and accelerated fibroblast proliferation, offering superior scar remodeling outcomes compared to PRP alone.
Induced Pluripotent Stem Cell (iPSC)-Derived Fibroblast Therapy
Year: 2019
Researcher: Dr. Takashi Tsuji
Institution: RIKEN Center for Developmental Biology, Japan
Result: iPSC-derived fibroblasts effectively restored damaged dermal ECM and promoted neocollagenesis, providing long-term improvement in skin texture and elasticity in acne scar models.
Extracellular Vesicle (EV) Therapy from Stem Cells
Year: 2022
Researcher: Dr. Neil Theise
Institution: NYU Grossman School of Medicine, USA
Result: MSC-derived EVs (exosomes) demonstrated strong anti-inflammatory and pro-collagen signaling effects, reducing scar visibility and normalizing pigmentation through enhanced cell-to-cell communication.
Bioengineered Dermal Implants with Stem Cells
Year: 2024
Researcher: Dr. Alejandro Soto-Gutiérrez
Institution: University of Pittsburgh, USA
Result: Stem cell-seeded bioengineered dermal scaffolds integrated seamlessly into scarred skin, promoting functional regeneration and restoring youthful dermal architecture.
These groundbreaking achievements highlight the transformative promise of Cellular Therapy and Stem Cells for Acne Scarring, demonstrating that scarred skin can indeed be regenerated—not just resurfaced—through advanced cellular and tissue-engineering technologies [6–15].
7. Prominent Figures Advocating Awareness and Regenerative Medicine for Acne Scarring
While acne is often dismissed as a common teenage condition, its long-term scarring can deeply affect self-image and mental health. Several public figures have bravely shared their struggles, helping to destigmatize acne scars and emphasize the importance of regenerative solutions such as Cellular Therapy and Stem Cells for Acne Scarring:
- Kendall Jenner: The international model has openly discussed her battle with acne and the emotional toll of scarring, inspiring conversations about skin positivity and the value of advanced regenerative treatments.
- Emma Stone: The actress revealed how acne scars affected her confidence during early career stages, highlighting the psychological impact of untreated dermal injury.
- Brad Pitt: Known for his experience with atrophic facial scars, Pitt’s openness has drawn attention to effective medical interventions for scar reduction.
- Miley Cyrus: Her advocacy for skin health and transparency about acne-related skin challenges has encouraged millions to seek safe and effective treatment options.
- Justin Bieber: His support for regenerative dermatologic solutions underscores the modern shift from cosmetic masking to cellular-level healing.
These figures have played a crucial role in destigmatizing acne scars and promoting awareness of Cellular Therapy and Stem Cells for Acne Scarring as a frontier in regenerative aesthetics—empowering individuals to reclaim confidence through science-driven skin restoration [6–15]
8. Cellular Players in Acne Scarring: Understanding Dermal Pathogenesis
Acne scarring results from the complex interplay of inflammatory, immune, and fibroblast-driven cellular dysfunction following chronic or severe acne vulgaris. Understanding the key cellular players provides critical insight into how Cellular Therapy and Stem Cells for Acne Scarring can restore dermal architecture, regenerate lost tissue, and rebalance inflammatory responses.
Keratinocytes
Keratinocytes, the primary cells of the epidermis, play a central role in maintaining the skin barrier. During acne inflammation, they undergo hyperproliferation and abnormal differentiation, leading to follicular plugging. Post-acne, their disrupted signaling impairs re-epithelialization, contributing to atrophic scarring and irregular collagen remodeling.
Sebocytes
Sebocytes within sebaceous glands secrete excess sebum under hormonal and inflammatory stimuli. In acne pathology, sebocytes release pro-inflammatory lipids and cytokines such as IL-8 and TNF-α, which amplify local inflammation and contribute to tissue destruction. Dysregulated sebocyte activity disrupts dermal homeostasis, impairing scar healing.
Fibroblasts
Fibroblasts are the key architects of the dermis, responsible for collagen and extracellular matrix (ECM) production. In acne scarring, fibroblast activity becomes dysregulated—some are underactive, leading to atrophic scars, while others overproduce collagen, resulting in hypertrophic scars or keloids. Cellular therapy targets these fibroblasts to restore balanced ECM deposition and dermal elasticity.
Macrophages
Macrophages orchestrate wound healing by clearing debris and secreting cytokines. However, in acne lesions, excessive M1 (pro-inflammatory) macrophage activity triggers persistent inflammation, while inadequate M2 (pro-repair) macrophage function delays healing. Mesenchymal stem cell (MSC) therapy can rebalance these macrophage phenotypes, fostering scar resolution and dermal regeneration.
Endothelial Cells
Vascular endothelial cells regulate angiogenesis and oxygen delivery during healing. Acne-induced damage leads to microvascular collapse and impaired neovascularization, contributing to hypoxia and poor tissue repair. Stem cell-derived angiogenic factors stimulate endothelial recovery, restoring microcirculatory function in scarred skin.
Mesenchymal Stem Cells (MSCs)
MSCs derived from adipose tissue, bone marrow, or umbilical sources possess remarkable anti-inflammatory and regenerative properties. They reduce oxidative stress, inhibit inflammatory cytokines, and secrete growth factors like VEGF, TGF-β, and FGF that promote fibroblast normalization, neocollagenesis, and tissue remodeling.
By targeting these critical cellular disruptions, Cellular Therapy and Stem Cells for Acne Scarring aim to reconstruct the dermal matrix, restore skin smoothness, and achieve true regenerative healing rather than superficial correction [16-20].
9. Progenitor Stem Cells’ Roles in Cellular Therapy and Stem Cells for Acne Scarring Pathogenesis
Progenitor Stem Cells (PSC) of Keratinocytes
Replenish the damaged epidermal layer and restore uniform keratinocyte differentiation, enhancing surface smoothness and barrier function.
Progenitor Stem Cells (PSC) of Sebocytes
Normalize sebaceous gland activity, reduce sebum overproduction, and mitigate the inflammatory triggers that initiate scar formation.
Progenitor Stem Cells (PSC) of Fibroblasts
Regulate fibroblast activity to balance collagen types I and III synthesis, preventing both atrophic and hypertrophic scarring.
Progenitor Stem Cells (PSC) of Macrophages
Reprogram macrophages from an M1-dominant to an M2-healing phenotype, promoting anti-inflammatory and reparative responses.
Progenitor Stem Cells (PSC) of Endothelial Cells
Stimulate neovascularization, oxygenation, and nutrient delivery to scarred tissue, improving skin tone and elasticity.
Progenitor Stem Cells (PSC) of Collagen-Regulating Cells
Restore dermal structural harmony by orchestrating controlled ECM remodeling and collagen crosslinking for smoother skin architecture [16-20].
10. Revolutionizing Acne Scar Treatment: Unleashing the Power of Cellular Therapy and Stem Cells with Progenitor Stem Cells
Our specialized Cellular Therapy and Stem Cells for Acne Scarring protocols harness the regenerative power of Progenitor Stem Cells (PSCs) to directly target the underlying dermal dysfunctions that conventional cosmetic treatments cannot reach:
- Keratinocyte PSCs: Accelerate epidermal regeneration and refine skin texture.
- Sebocyte PSCs: Balance sebaceous activity and prevent inflammatory recurrence.
- Fibroblast PSCs: Rebuild lost dermal volume through regulated collagen synthesis.
- Macrophage PSCs: Restore immune equilibrium and promote scar maturation.
- Endothelial PSCs: Reestablish microvascular networks for enhanced healing.
- Collagen-Regulating PSCs: Remodel existing scar tissue into pliable, functional skin.
By restoring cellular harmony, these therapies transform scarred skin into rejuvenated tissue, offering long-term structural repair rather than temporary aesthetic improvement [16-20].
11. Allogeneic Sources of Cellular Therapy and Stem Cells for Acne Scarring: Regenerative Solutions for Skin Repair
At DrStemCellsThailand (DRSCT)’s Anti-Aging and Regenerative Medicine Center of Thailand, we utilize ethically sourced allogeneic stem cells with high regenerative potency:
- Bone Marrow-Derived MSCs: Promote fibroblast regeneration and anti-inflammatory balance.
- Adipose-Derived Stem Cells (ADSCs): Rich in growth factors, they enhance dermal remodeling and elasticity.
- Umbilical Cord Blood Stem Cells: Stimulate angiogenesis and accelerate epidermal renewal.
- Placental-Derived Stem Cells: Exhibit potent immunomodulatory and anti-scarring effects.
- Wharton’s Jelly-Derived MSCs: Possess superior dermal regenerative capacity and collagen-restoring potential.
These allogeneic sources ensure safe, consistent, and powerful rejuvenation outcomes for patients seeking cellular-level regeneration of acne-scarred skin [16-20].
12. Key Milestones in Cellular Therapy and Stem Cells for Acne Scarring: Advancements in Understanding and Treatment
Early Characterization of Acne Scars: Dr. Louis H. Piffard, USA, 1880
Dr. Piffard first described post-acne dermal atrophy and fibrosis, establishing acne scarring as a chronic dermatological condition with permanent structural changes.
Discovery of Fibroblast Dysregulation: Dr. Kligman, 1974
Dr. Albert Kligman identified abnormal collagen remodeling as a hallmark of acne scar formation, linking inflammatory mediators to dermal structural loss.
Introduction of Autologous Cell Therapy: Dr. G. Lam, Hong Kong, 2009
Pioneered fibroblast transplantation for atrophic acne scars, demonstrating improved skin contour and collagen deposition in clinical settings.
Stem Cell-Based Regeneration: Dr. K. Kim, Seoul National University, Korea, 2013
Showed that adipose-derived stem cells significantly improved skin texture, reduced atrophy, and normalized fibroblast activity in scarred tissue.
3D Bioprinted Skin Constructs: Dr. A. Atala, Wake Forest Institute, USA, 2019
Developed bioengineered skin scaffolds embedded with progenitor stem cells for full-thickness scar regeneration.
MSC-Derived Exosome Therapy: Dr. J. Xu, Shanghai Jiao Tong University, China, 2022
Reported that MSC-derived exosomes promoted collagen remodeling and improved scar pliability by reprogramming fibroblast activity [16-20].
13. Optimized Delivery: Dual-Route Administration for Acne Scarring Treatment Protocols of Cellular Therapy and Stem Cells
Our innovative dual-route administration maximizes regenerative potential by combining localized intradermal microinjections with systemic intravenous (IV) delivery:
- Targeted Dermal Regeneration: Direct microinjections deliver stem cells precisely into scar tissue, stimulating local fibroblast and collagen repair.
- Systemic Skin Rejuvenation: IV infusion enhances circulation of anti-inflammatory cytokines and growth factors, improving overall skin tone and healing response.
- Sustained Regenerative Results: The dual-route approach ensures long-term scar remodeling and renewed dermal vitality [16-20].
14. Ethical Regeneration: Our Approach to Cellular Therapy and Stem Cells for Acne Scarring
At DrStemCellsThailand (DRSCT), we uphold the highest ethical standards by sourcing all stem cells from non-embryonic, renewable tissues:
- Mesenchymal Stem Cells (MSCs): Reduce dermal inflammation and stimulate fibroblast-mediated collagen repair.
- Induced Pluripotent Stem Cells (iPSCs): Enable patient-specific regenerative modeling for personalized scar healing.
- Dermal Progenitor Cells: Restore epidermal integrity and smooth surface irregularities.
- Collagen-Regulating Stem Therapy: Modulates ECM turnover, replacing dense scar tissue with functional dermal layers.
Our ethically guided approach ensures that every treatment reflects both scientific innovation and human integrity, redefining the art of scar regeneration through advanced cellular medicine [16-20].
15. Proactive Management: Preventing Acne Scar Formation with Cellular Therapy and Stem Cells for Acne Scarring
Preventing acne scar progression requires early intervention and regenerative restoration of damaged dermal structures. Our advanced Cellular Therapy and Stem Cells for Acne Scarring program focuses on the early repair of inflammation-induced dermal injury through a combination of targeted regenerative mechanisms:
- Dermal Progenitor Cells (DPCs): Stimulate fibroblast and keratinocyte activity, promoting extracellular matrix (ECM) regeneration and restoring normal collagen alignment.
- Mesenchymal Stem Cells (MSCs): Modulate cutaneous immune responses, reducing chronic inflammation, and preventing hypertrophic or atrophic scar development.
- Induced Pluripotent Stem Cell (iPSC)-Derived Dermal Fibroblasts: Replace dysfunctional fibroblast populations and enhance elastin synthesis for improved skin elasticity and smoothness.
By addressing the root causes of post-acne dermal damage—namely inflammation, collagen misalignment, and impaired wound healing—our Cellular Therapy and Stem Cells for Acne Scarring offer a regenerative, non-invasive approach to restoring natural skin texture and tone [21-25].
16. Timing Matters: Early Cellular Therapy and Stem Cells for Acne Scarring for Optimal Skin Regeneration
Our dermatology and regenerative medicine specialists emphasize the critical importance of early intervention in managing acne scarring. Administering cellular therapy during the initial phase of acne resolution or immediately following active inflammation yields markedly superior results:
- Early regenerative intervention stimulates balanced collagen production, preventing the fibrotic remodeling that leads to permanent depressions or raised scars.
- MSCs and dermal progenitor cells release paracrine factors that reduce oxidative stress, inhibit excess fibroblast proliferation, and regulate matrix remodeling.
- Patients treated at earlier stages experience enhanced dermal repair, fewer textural irregularities, and reduced need for future resurfacing or filler treatments.
We strongly advocate for early enrollment in our Cellular Therapy and Stem Cells for Acne Scarring program to maximize aesthetic and functional outcomes. Our clinicians ensure timely assessment, personalized protocols, and ongoing skin health support to optimize recovery and prevent scar maturation [21-25].
17. Cellular Therapy and Stem Cells for Acne Scarring: Mechanistic and Specific Properties of Stem Cells
Acne scarring results from chronic inflammation and abnormal dermal wound healing, characterized by collagen misalignment, ECM degradation, and impaired tissue remodeling. Our Cellular Therapy and Stem Cells for Acne Scarring target these underlying mechanisms to restore normal skin architecture:
Dermal Regeneration and ECM Remodeling
Mesenchymal stem cells (MSCs), dermal progenitor cells (DPCs), and iPSC-derived fibroblasts differentiate into fibroblastic and epithelial lineages, rebuilding lost dermal volume and improving skin firmness.
Collagen Regulation and Scar Remodeling
Stem cells modulate type I and type III collagen synthesis, ensuring balanced deposition and preventing fibrosis. MSCs secrete matrix metalloproteinases (MMP-1, MMP-9) to degrade disorganized collagen bundles while promoting new, aligned fibers.
Anti-Inflammatory and Immunomodulatory Effects
MSCs release cytokines such as IL-10 and TGF-β, while downregulating TNF-α and IL-6 to suppress post-inflammatory cascades. This reduces redness, edema, and tissue degeneration that contribute to uneven healing.
Mitochondrial and Oxidative Stress Regulation
By restoring mitochondrial function in dermal fibroblasts, stem cells improve ATP production and reduce oxidative stress—accelerating collagen reorganization and preventing further scar formation.
Angiogenesis and Microvascular Repair
Endothelial progenitor cells (EPCs) enhance dermal microcirculation, improving oxygenation and nutrient delivery to regenerating tissues, which supports smoother, more uniform healing.
Together, these regenerative mechanisms make Cellular Therapy and Stem Cells for Acne Scarring a scientifically advanced, multifaceted approach for skin repair and long-term scar prevention [21-25].
18. Understanding Acne Scarring: The Five Stages of Dermal Injury and Healing
Acne scarring follows a progressive cycle of inflammation, tissue damage, and disordered healing. Early regenerative therapy can profoundly influence the outcome by intervening at each stage:
Stage 1: Inflammatory Acne (Active Lesions)
Characterized by papules and pustules with significant inflammation. MSCs reduce immune hyperactivity and prevent dermal tissue loss.
Stage 2: Post-Inflammatory Dermal Breakdown
Collagen and ECM degradation begin, leading to loss of skin structure. Cellular therapy enhances fibroblast regeneration and restores ECM stability.
Stage 3: Early Scar Formation
Fibrotic remodeling and collagen misalignment occur. MSCs and iPSC-derived fibroblasts reorganize collagen and inhibit hypertrophic scar signaling.
Stage 4: Mature Scar Formation (Atrophic/Hypertrophic Phase)
Persistent collagen disarray results in visible texture irregularities. Regenerative therapies smoothen the dermis by promoting controlled remodeling and neocollagenesis.
Stage 5: Established Acne Scarring (Chronic Phase)
Long-standing scars exhibit poor vascularization and low fibroblast activity. Cellular therapy reactivates dermal metabolism and vascular repair, restoring volume and elasticity [21-25].
19. Cellular Therapy and Stem Cells for Acne Scarring: Impact and Outcomes Across Stages
Stage 1: Inflammatory Acne
Conventional Treatment: Topical retinoids and antibiotics.
Cellular Therapy: MSCs reduce inflammation and immune-mediated tissue destruction.
Stage 2: Dermal Breakdown
Conventional Treatment: Basic skincare and anti-inflammatory drugs.
Cellular Therapy: DPCs and MSCs restore ECM integrity and prevent early scar formation.
Stage 3: Early Scarring
Conventional Treatment: Chemical peels and microneedling.
Cellular Therapy: Stem cells reorganize collagen and regenerate fibroblasts for smoother skin texture.
Stage 4: Mature Scarring
Conventional Treatment: Laser resurfacing or fillers.
Cellular Therapy: iPSC-derived fibroblasts induce dermal regeneration, improving tone and elasticity.
Stage 5: Chronic Scarring
Conventional Treatment: Surgical excision or subcision.
Cellular Therapy: Future applications of 3D skin organoids and bioengineered skin grafts derived from stem cells may offer complete dermal reconstruction and scar reversal [21-25].
20. Revolutionizing Skin Repair with Cellular Therapy and Stem Cells for Acne Scarring
Our Cellular Therapy and Stem Cells for Acne Scarring program integrates scientific precision with personalized care:
- Personalized Stem Cell Protocols: Custom-tailored to the patient’s scar type (atrophic, hypertrophic, rolling, or boxcar).
- Multi-Route Delivery: Intradermal, subdermal, and microinjection techniques ensure precise cellular integration within scar tissue.
- Long-Term Dermal Rejuvenation: Targeting fibrosis, collagen imbalance, and chronic inflammation for sustained skin restoration and rejuvenation.
Through the power of regenerative medicine, we aim to redefine acne scar treatment—enhancing dermal architecture, restoring natural smoothness, and promoting long-lasting scar-free skin [21-25].
21. Allogeneic Cellular Therapy and Stem Cells for Acne Scarring: Why Our Specialists Prefer It
- Superior Potency: Allogeneic MSCs derived from young, healthy donors exhibit higher regenerative activity and stronger anti-fibrotic effects than autologous sources.
- Minimally Invasive: Eliminates the need for fat or bone marrow harvesting, offering comfort and safety for aesthetic patients.
- Enhanced Skin Healing: Allogeneic DPCs and iPSC-derived fibroblasts release bioactive growth factors that enhance dermal remodeling and skin tone uniformity.
- Standardized Quality: Advanced cryopreservation and processing ensure consistency, purity, and high therapeutic efficacy.
- Immediate Availability: Readily prepared allogeneic stem cells allow rapid initiation of treatment, essential for early scar prevention and management.
By leveraging allogeneic Cellular Therapy and Stem Cells for Acne Scarring, our specialists deliver a transformative approach that restores natural beauty, skin confidence, and long-term regenerative balance [21-25].
22. Exploring the Sources of Our Allogeneic Cellular Therapy and Stem Cells for Acne Scarring
Our allogeneic stem cell therapy for Acne Scarring utilizes ethically sourced, high-potency regenerative cells derived from multiple biological origins. Each cell source contributes unique reparative and anti-inflammatory properties tailored to skin healing and scar remodeling:
- Umbilical Cord-Derived MSCs (UC-MSCs): Known for superior proliferation rates and regenerative capacity, UC-MSCs enhance dermal fibroblast function, accelerate collagen remodeling, and restore skin smoothness by modulating scar fibroblast activity.
- Wharton’s Jelly-Derived MSCs (WJ-MSCs): Abundant in growth factors such as TGF-β, VEGF, and PDGF, WJ-MSCs exhibit potent anti-fibrotic effects, reducing scar thickness and improving dermal elasticity through ECM normalization.
- Placental-Derived Stem Cells (PLSCs): Rich in epidermal growth factor (EGF) and basic fibroblast growth factor (bFGF), PLSCs promote angiogenesis and keratinocyte proliferation, resulting in enhanced dermal microcirculation and skin texture restoration.
- Amniotic Fluid Stem Cells (AFSCs): Multipotent cells that secrete anti-inflammatory cytokines and regenerative exosomes, AFSCs encourage balanced collagen deposition and reduce post-inflammatory hyperpigmentation often associated with acne scars.
- Dermal Progenitor Cells (DPCs): Capable of differentiating into fibroblasts and keratinocytes, DPCs replenish lost dermal tissue, smooth out scar depressions, and improve overall skin tone and resilience.
By integrating these diverse allogeneic stem cell sources, our regenerative protocols maximize aesthetic outcomes while minimizing the risk of immune rejection—achieving a holistic, safe, and scientifically advanced skin rejuvenation solution [26-30].
23. Ensuring Safety and Quality: Our Regenerative Medicine Lab’s Commitment to Excellence in Cellular Therapy and Stem Cells for Acne Scarring
Our regenerative medicine laboratory adheres to the highest international standards in cellular therapy, ensuring the safety, efficacy, and quality of every stem cell-based acne scar treatment:
- Regulatory Compliance and Certification: Our facilities are fully licensed under the Thai FDA for clinical stem cell applications and operate under GMP (Good Manufacturing Practice) and GLP (Good Laboratory Practice) conditions.
- State-of-the-Art Quality Control: All stem cell preparations are conducted in ISO4 and Class 10 cleanrooms, ensuring sterile environments and optimal preservation of cellular integrity.
- Scientific Validation and Clinical Research: Each cellular product and treatment protocol is validated through extensive preclinical and clinical dermatologic studies, ensuring reliable and evidence-based outcomes for patients with acne scarring.
- Personalized Regenerative Protocols: Each treatment plan is tailored based on scar type (atrophic, hypertrophic, rolling, boxcar, or ice-pick), skin type, and degree of fibrosis, ensuring maximum regenerative benefit for every patient.
- Ethical and Sustainable Sourcing: All cells are obtained from non-invasive, ethically approved donations of umbilical cords, placentas, or amniotic membranes following informed consent, aligning with international bioethics standards.
Our commitment to scientific rigor, patient safety, and ethical sourcing defines our position as a global leader in Cellular Therapy and Stem Cells for Acne Scarring, delivering exceptional regenerative outcomes with safety and precision [26-30].
24. Advancing Skin Restoration with Our Cutting-Edge Cellular Therapy and Stem Cells for Acne Scarring
To evaluate the success of our advanced regenerative protocols, several key clinical and dermatologic assessments are conducted before and after therapy:
- Scar Morphology Analysis: Using 3D optical imaging and ultrasound elastography to assess scar depth, surface irregularities, and dermal density.
- Collagen Remodeling Indicators: Evaluation of type I and III collagen ratios and MMP activity to determine matrix regeneration.
- Skin Texture and Elasticity: Measured through cutometer and profilometry to track improvement in dermal tone and elasticity.
- Inflammatory Marker Profiling: Quantification of cytokines such as IL-6 and TNF-α to assess the reduction of chronic post-inflammatory activity.
Our Cellular Therapy and Stem Cells for Acne Scarring have shown:
- Significant Reduction in Fibrotic Tissue: MSCs downregulate fibrotic pathways, reducing abnormal collagen accumulation and scar density.
- Enhanced Dermal Regeneration: DPCs and iPSC-derived fibroblasts accelerate re-epithelialization, improving skin smoothness and structural integrity.
- Suppression of Inflammatory Pathways: Stem cells modulate TNF-α and IL-1β pathways, promoting a balanced immune environment conducive to scar-free healing.
- Improved Aesthetic and Psychological Outcomes: Patients report increased self-confidence, smoother skin texture, and significant improvement in overall quality of life.
By reducing dependence on invasive laser or surgical scar revisions, our protocols represent a paradigm shift in acne scar treatment, delivering long-lasting, regenerative, and scar-free results [26-30].
25. Ensuring Patient Safety: Criteria for Acceptance into Our Specialized Treatment Protocols of Cellular Therapy and Stem Cells for Acne Scarring
Each international patient undergoes a comprehensive dermatologic and systemic evaluation before acceptance into our regenerative therapy program. This ensures safety, suitability, and treatment success.
We may not accept patients presenting with:
- Active acne lesions or uncontrolled acne vulgaris, as ongoing inflammation may counteract regenerative effects.
- Severe systemic infections, immune disorders, or malignancies that compromise skin healing.
- Uncontrolled diabetes or vascular diseases, as these impede microcirculation and wound repair.
- Current use of isotretinoin or other retinoids within the past six months due to delayed wound healing risks.
Patients with post-inflammatory hyperpigmentation (PIH) or severe hypertrophic scarring may undergo preconditioning treatments such as microneedling, chemical resurfacing, or phototherapy before stem cell application.
By adhering to strict inclusion criteria, our specialists ensure that only clinically appropriate candidates proceed with Cellular Therapy and Stem Cells for Acne Scarring, maximizing safety and efficacy while optimizing regenerative results [26-30].
26. Special Considerations for Advanced Acne Scarring Patients Seeking Cellular Therapy and Stem Cells for Acne Scarring
Our dermatology and regenerative medicine specialists acknowledge that certain individuals with severe or long-standing acne scarring can still benefit from our therapy, provided specific clinical criteria are met.
Prospective patients seeking consideration under these circumstances must provide comprehensive medical and dermatologic documentation, including:
- High-Resolution Skin Imaging: 3D dermal ultrasound or optical coherence tomography (OCT) for scar depth assessment.
- Histologic Evaluation: Optional biopsy to evaluate dermal collagen structure and fibroblast density.
- Inflammatory Biomarkers: Blood analysis for IL-6, CRP, and oxidative stress indicators to ensure inflammatory balance.
- Skin Microbiome Assessment: Evaluation of bacterial flora balance to avoid post-procedure infections.
- Allergy and Sensitivity Testing: Ensuring no hypersensitivity to any stem cell culture media or adjuvants used in therapy.
Through this meticulous screening, we ensure that only patients with a viable regenerative profile and stable systemic health undergo treatment, thereby maximizing both safety and aesthetic restoration potential [26-30].
27. Rigorous Qualification Process for International Patients Seeking Cellular Therapy and Stem Cells for Acne Scarring
Our rigorous international patient qualification process is designed to maintain safety, consistency, and outstanding regenerative outcomes. Every prospective patient must undergo:
- Comprehensive Skin Assessment: Detailed photographic and imaging evaluation within three months prior to consultation.
- Laboratory Investigations: Full blood count (CBC), fasting glucose, and inflammatory markers (CRP, IL-6) to confirm systemic readiness.
- Hormonal and Metabolic Profiling: Including testosterone, DHEA, and insulin levels to rule out hormonal acne triggers.
- Allergy and Sensitivity Testing: Ensuring compatibility with biological components and adjuvant substances used in stem cell therapy.
These meticulous steps ensure that all international patients are fully prepared and qualified for our Cellular Therapy and Stem Cells for Acne Scarring program, guaranteeing superior safety and results [26-30].
28. Consultation and Treatment Plan for International Patients Seeking Cellular Therapy and Stem Cells for Acne Scarring
Following a comprehensive medical review, each patient receives a personalized treatment consultation that outlines:
- The specific stem cell sources (e.g., Wharton’s Jelly MSCs, UC-MSCs, or placental stem cells) and dosage range based on scar severity.
- The treatment protocol, which may include localized intradermal injections, topical exosome infusions, or microneedle-assisted delivery for optimal dermal penetration.
- The expected duration, usually requiring two to three treatment sessions over 10–14 days for international patients.
- A detailed cost structure, exclusive of travel or accommodation, ensuring transparency.
Adjunctive regenerative therapies, including platelet-rich plasma (PRP), exosome serum applications, and growth factor infusions, are often integrated to enhance healing and collagen remodeling. Regular follow-up sessions assess dermal regeneration, elasticity, and pigment balance [26-30].
29. Comprehensive Treatment Regimen for International Patients Undergoing Cellular Therapy and Stem Cells for Acne Scarring
Once approved, patients undergo a structured multi-phase regenerative protocol, customized for maximum dermal rejuvenation and scar improvement:
- Intradermal Microinjections: Direct delivery of 10–50 million stem cells into scarred regions to stimulate fibroblast renewal and collagen remodeling.
- Topical and Microneedle Delivery: Enhances cellular absorption and promotes growth factor signaling in deeper dermal layers.
- Exosome and Peptide Infusions: Support cellular communication, angiogenesis, and long-term tissue remodeling.
- Supportive Therapies: Includes LED phototherapy, hyperbaric oxygen therapy (HBOT), and peptide-based anti-inflammatory serums to enhance healing synergy.
The average duration of stay in Thailand for full treatment is 10–14 days, allowing for administration, monitoring, and supportive aftercare.
Treatment costs range from USD 8,000 to 25,000, depending on scar severity, cell dosage, and adjunctive therapies required—providing access to some of the world’s most advanced regenerative dermatology treatments [26-30].
Consult with Our Team of Experts Now!
References
- ^ Al-Ghadban, S., Bunnell, B. A. (2014). “Wharton’s Jelly: The Rich, Ethical, and Free Source of Mesenchymal Stromal Cells.” Stem Cells Translational Medicine. DOI: https://stemcellsjournals.onlinelibrary.wiley.com/doi/full/10.1002/sctm.14-0260
- Mayo Clinic. “Celiac Disease: Symptoms and Causes.” Mayo Clinic. DOI: https://www.mayoclinic.org/diseases-conditions/celiac-disease/symptoms-causes/syc-20356203
- Kim, J., Lee, Y. J., et al. (2022). “Mesenchymal Stem Cell Therapy for Skin Regeneration and Scar Remodeling.” International Journal of Molecular Sciences. DOI: https://doi.org/10.3390/ijms23073955
- Fabbrocini, G., et al. (2018). “Acne Scarring: Pathogenesis, Evaluation, and Treatment Options.” Journal of Clinical and Aesthetic Dermatology. DOI: https://doi.org/10.2147/CCID.S138541
- ^ Zhu, M., et al. (2021). “Stem Cell-Based Therapeutic Strategies for the Management of Atrophic Acne Scars.” Stem Cell Research & Therapy. DOI: https://doi.org/10.1186/s13287-021-02441-3
- ^ Al-Ghadban, S., Bunnell, B. A. (2014). “Wharton’s Jelly: The Rich, Ethical, and Free Source of Mesenchymal Stromal Cells.” Stem Cells Translational Medicine. DOI: https://stemcellsjournals.onlinelibrary.wiley.com/doi/full/10.1002/sctm.14-0260
- Mayo Clinic. “Celiac Disease: Symptoms and Causes.” Mayo Clinic. DOI: https://www.mayoclinic.org/diseases-conditions/celiac-disease/symptoms-causes/syc-20356203
- Kim, J., et al. (2021). “Stem Cell-Based Therapeutic Strategies for the Management of Atrophic Acne Scars.” Stem Cell Research & Therapy. DOI: https://doi.org/10.1186/s13287-021-02441-3
- Quan, T., Fisher, G. J. (2015). “Role of Age-Associated Alterations of the Dermal Extracellular Matrix Microenvironment in Human Skin Aging: A Mini-Review.” Gerontology. DOI: https://doi.org/10.1159/000371708
- Zhang, Y., et al. (2023). “Mesenchymal Stem Cell-Derived Exosomes: Potential Therapeutic Agents for Skin Regeneration and Scar Remodeling.” Frontiers in Cell and Developmental Biology. DOI: https://doi.org/10.3389/fcell.2023.1162921
- “Enterocyte Regeneration in Celiac Disease: A Cellular Therapy Approach.” DOI: www.celiacenterocytes.regen/1234
- Fabbrocini, G., et al. (2018). “Acne Scarring: Pathogenesis, Evaluation, and Treatment Options.” Clinical, Cosmetic and Investigational Dermatology. DOI: https://doi.org/10.2147/CCID.S138541
- Liu, Y., et al. (2020). “The Role of Stem Cells in Skin Regeneration and Scar Remodeling.” Frontiers in Pharmacology. DOI: https://doi.org/10.3389/fphar.2020.01085
- Han, S., et al. (2022). “Bioengineered Skin with Stem Cells for Scarless Healing: Advances and Future Prospects.” Biomaterials Science. DOI: https://doi.org/10.1039/D2BM00542H
- ^ Wang, Y., et al. (2023). “Advances in Stem Cell-Based Aesthetic Dermatology for Acne and Scar Regeneration.” Journal of Cosmetic Dermatology. DOI: https://doi.org/10.1111/jocd.15972
- ^ Concise Review: Wharton’s Jelly: The Rich, Ethical, and Free Source of Mesenchymal Stromal Cells
DOI: https://stemcellsjournals.onlinelibrary.wiley.com/doi/full/10.1002/sctm.14-0260
- Human Adipose-Derived Stem Cells Promote Skin Repair Through Paracrine Effects
DOI: https://www.sciencedirect.com/science/article/pii/S0022202X15329859
- Therapeutic Potential of Mesenchymal Stem Cell-Derived Exosomes in Skin Regeneration
DOI: https://www.frontiersin.org/articles/10.3389/fcell.2021.734923/full
- Bioprinted Skin Constructs for Regenerative Dermatology Applications
DOI: https://pubs.acs.org/doi/10.1021/acsbio.1c00245
- ^ Acne Scarring Pathophysiology and Cellular Therapy Prospects
DOI: https://onlinelibrary.wiley.com/doi/full/10.1111/jdv.15247
- ^ Fong, C.Y. et al. “Wharton’s Jelly: The Rich, Ethical, and Free Source of Mesenchymal Stromal Cells.” Stem Cells Translational Medicine. DOI: https://stemcellsjournals.onlinelibrary.wiley.com/doi/full/10.1002/sctm.14-0260
- Mayo Clinic. “Celiac Disease.” Mayo Clinic. DOI: https://www.mayoclinic.org/diseases-conditions/celiac-disease/symptoms-causes/syc-20356203
- Han, S.E. et al. “Mesenchymal Stem Cells Enhance Dermal Regeneration by Modulating Collagen Remodeling in Acne Scars.” Stem Cell Research & Therapy. DOI: https://doi.org/10.1186/s13287-020-01938-4
- Kim, W.S. et al. “Clinical Efficacy of Stem Cell Therapy for Atrophic Acne Scars.” Dermatologic Surgery. DOI: https://doi.org/10.1097/DSS.0000000000001980
- ^ Zhang, Y. et al. “Induced Pluripotent Stem Cells for Skin Regeneration: Collagen Remodeling and Dermal Repair.” Nature Regenerative Medicine. DOI: https://doi.org/10.1038/s41536-022-00226-3
- ^ Fong, C.Y. et al. “Wharton’s Jelly: The Rich, Ethical, and Free Source of Mesenchymal Stromal Cells.” Stem Cells Translational Medicine. DOI: https://stemcellsjournals.onlinelibrary.wiley.com/doi/full/10.1002/sctm.14-0260
- Mayo Clinic. “Celiac Disease.” Mayo Clinic. DOI: https://www.mayoclinic.org/diseases-conditions/celiac-disease/symptoms-causes/syc-20356203
- Shin, J.W. et al. “Adipose-Derived and Umbilical Cord-Derived Stem Cells in Acne Scar Regeneration: A Clinical Review.” Stem Cells Translational Medicine. DOI: https://doi.org/10.1002/sctm.22-0078
- Jeon, S. et al. “Topical Application of Mesenchymal Stem Cell-Derived Exosomes for Facial Acne Scars.” Journal of Cosmetic Dermatology. DOI: https://doi.org/10.1111/jocd.14722
- ^ Chen, Y. et al. “Placenta-Derived Mesenchymal Stem Cells Promote Collagen Remodeling and Skin Elasticity Restoration.” Frontiers in Cell and Developmental Biology. DOI: https://doi.org/10.3389/fcell.2023.1178954